Screening Muscles, Fascia, Joints for Chronic GVHD
Below are clinical manifestations in a patient's muscles, fascia, and joints that are potential early indicators of chronic GVHD. If GVHD is suspected, timely collaboration with the patient's transplant center is recommended to confirm the diagnosis and to develop and evaluate a treatment plan.
These guidelines are based on published diagnostic criteria from the National Institutes of Health (NIH) Consensus Development Project on chronic GVHD. [1,2,3]
Clinical Examination
- Palpation for areas of thickening, tightening, shortening of muscles or fascia; muscle tenderness
- Evaluate range of motion
- Muscle strength testing
- Inspection for signs of edema or peau d'orange skin changes
- Visual inspection for grooving, ridging
Diagnostic Testing
- Creatinine kinase
- Aldolase
- Electromyography
Patient-Reported Symptoms and Signs
- Muscle cramps
- Muscle pain
- Muscle weakness
- Joint stiffness
- Restricted range of motion
- Tightened muscles, tendons, and fascia
- Contractures
Possible Manifestations
Fasciitis
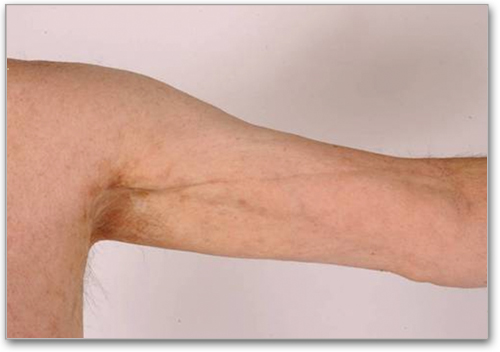
Stiffness, restricted range of motion
Joint stiffness or contractures (secondary to fasciitis or sclerosis)
Groove sign, dimpling
Myositis or polymyositis*
Muscle tenderness and elevated muscle enzymes. Evaluate with electromyography and measurement of creatinine phosphokinase and aldolase. Muscle/sural nerve biopsies should be considered in the absence of other manifestations of GVHD to rule out other causes of myositis
Edema**
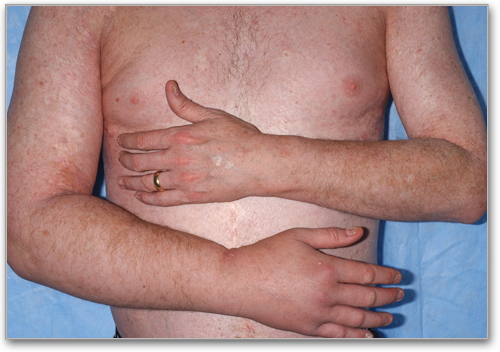
Present in extremities, with or without erythema and peau d'orange skin
Muscle Cramps**
May be present with increased muscle enzymes
Arthralgia or Arthritis**
Uncommon, occasionally associated with the presence of autoantibodies
Notes
* Distinctive but insufficient alone to establish an unequivocal diagnosis of chronic GVHD without further testing or additional organ involvement
** Rare, controversial, or non-specific features of chronic GVHD
*** Common in both acute and chronic GVHD
References
- Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Response Criteria Working Group Report. Biol Blood Marrow Transplant. 2015; 21(3): 389-401.
- Lee SJ, Wolff D, Kitko C, et al. Measuring therapeutic response in chronic graft-versus-host disease. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. The 2014 Response Criteria Working Group Report. Biol Blood Marrow Transplant. 2015; 21(6): 984-999.
- These guidelines have been developed by the National Marrow Donor Program® (NMDP)/Be The Match® in consultation with Sandra A. Mitchell, CRNP, MScN, AOCN; National Institutes of Health Clinical Center; and Steven Z. Pavletic, M.D.; National Cancer Institute, National Institutes of Health, Bethesda, Md. The information in this document does not represent the official position of the NIH or the U.S. Government.
Additional review from:
- Dennis L. Confer, M.D., NMDP/Be The Match, Minneapolis, Minn.
- Linda J. Burns, M.D., NMDP/Be The Match, Minneapolis, Minn.
- Madan Jagasia, M.D., Vanderbilt University Medical Center, Nashville, Tenn.
- Stephanie J. Lee, M.D., Fred Hutchinson Cancer Research Center, Seattle, Wash.
Text adapted from reports of the NIH Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease from Biology of Blood and Marrow Transplantation by American Society for Blood and Marrow Transplantation. Reproduced with permission of Elsevier, Inc.
Photo Credits
- Photos/ Keratosis Pilaris; Lichen Planus-like; Hypopigmentation; Sclerosis; Erosion; Maculopapular: Maria L. Turner, M.D.; Edward W.Cowen, M.D.; Dermatology Branch, National Cancer Institute, NIH, Bethesda, Md.
- Photos/ Poikiloderma; Morphea; Lichen Planus-like; Lichen Sclerosus-like; Hyperpigmentation; Sclerosis; Nail dystrophy; Alopecia; Edema: Edward W. Cowen, M.D.; Dermatology Branch, National Cancer Institute, NIH, Bethesda, Md.
- Photos/ Lichen planus; Mucoceles; Erythema: Mark M. Schubert, D.D.S., M.S.D.; Fred Hutchinson Cancer Research Center, Seattle, Wash.
- Photo/ Keratoconjunctivitis: Mary E.D. Flowers, M.D.; University of Washington, Seattle, Wash.
- Photo/ Blepharitis: Janine A. Smith, M.D.; National Eye Institute, NIH, Bethesda, Md.
All photos used with permission.