Early HLA Typing for Improved Patient Outcomes
HLA Today is available to community hematology/oncology practices at no cost
When allogeneic hematopoietic cell transplantation (HCT) may be indicated for your patient, the road to transplant is complex. Identifying a suitable matched donor can be a major barrier.
Human leukocytic antigen (HLA) typing and cytogenetic testing at your practice at the time of diagnosis is a critical first step to removing that barrier. This allows the process for finding a related or unrelated donor to begin as early as possible, even before your patient has a transplant consultation.
Finding a suitable donor early means your patient is more likely to have HCT at the optimal time, which can lead to better outcomes for your patient.
That's where HLA Today comes in.
Our HLA Today program removes barriers to HLA typing if your hematology/oncology practice:
- does not have access to HLA typing or
- does not have a process in place to conduct HLA typing or
- cannot claim insurance reimbursement for typing
The program provides no-cost HLA typing for your patients and their family members at the time of diagnosis. You do not need to file insurance paperwork or wait for approvals for HLA typing. We cover the cost for patients and their relatives.
You can help your patients get to transplant sooner and more efficiently when you use HLA Today.
Here’s how the process works.
- Order free buccal swab kits
- Have kits available for newly diagnosed patients
- Swab your patient at diagnosis
(you can also request a swab kit and intermediate resolution HLA typing for family members) - Send the completed kit to us for free high-resolution patient HLA typing
- Receive donor match report and patient HLA typing (family member HLA typing results arrive separately)
- Use the information to make critical treatment decisions with your patient and a transplant center as early as possible
- Refer appropriate patients for consultation
Early HLA typing and referral allows treatment barriers to be addressed sooner
When HLA typing occurs at diagnosis, the process of finding an HLA matched related or unrelated donor for allogeneic HCT can start early. This can increase the likelihood your patient can undergo transplant at the optimal time.
That is important
because finding a suitable donor can take time, especially if the preliminary search report shows a patient has an HLA type not commonly found on the registry. Family member HLA typing offered through HLA Today will also let you know quickly if a relative is a match.
No matter the search report prognosis, we encourage you to have a conversation with a transplant center as early as possible.
If
your patient does not have a fully matched related donor, unrelated donor or cord blood unit available, early HLA typing allows the transplant center to evaluate alternative options. This could include a donor with a lower degree of HLA match or a
haploidentical related donor.
In addition, your patient’s care team will have more time to address any non-medical barriers to transplant. For example, issues with insurance or caregiver support.

“We are big on refer, refer, refer. The earlier the better. We use HLA Today so our patients who come in for induction are getting swabbed. We use HLA Today to get their families swabbed. And we open up the conversation about transplant all during induction. It’s made a big difference how we can engage with them about transplant at the community level and the patients and their families can start to prepare for what that looks like.”
Courtney Bellomo, MD
Hematologist
Director of Stem Cell Therapy
Hematology Lead CAR-T and Cellular Therapy
New York Oncology Hematology
Research shows clinical benefit of early HLA typing, rapid donor identification and early referral for HCT consultation
We’re offering HLA Today to hematologists/oncologists because research has shown you can increase the likelihood your patient will have better treatment outcomes by conducting HLA typing and cytogenetic testing at the time of diagnosis.1
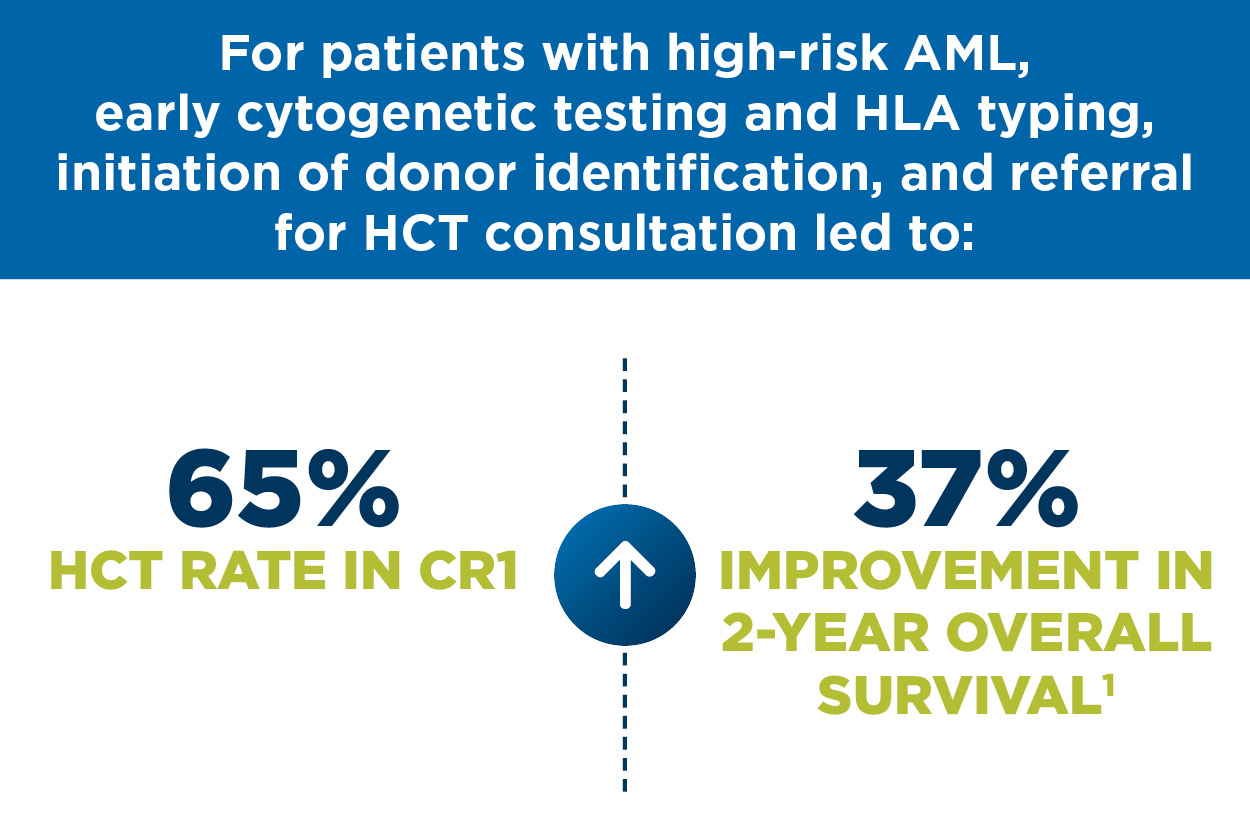
When you follow these standards of care, patients are more likely to have HCT in early-stage disease.
Studies on HCT revealed that patients who receive transplant in earlier disease stages have better outcomes than those transplanted in more
advanced disease stages.2
Learn more about the results of the research HLA Today is based on >>
Get started with HLA Today
HLA Today is available for any patient newly diagnosed with a condition for which HCT may be indicated.
The information you receive supports you in making critical treatment decisions with your patient as early as possible.
Additional Resources to Support Your Practice
- Transplant Consultation Timing Guidelines: Identify appropriate HCT referral timing based on disease characteristics
- Post-Transplant Care Recommendations: Deliver the specialized care your patients need when they return to your care
- Services for Patients: Access education, financial assistance and clinical trial support to help your patients overcome treatment obstacles
References
- Pagel JM, Othus M, Garcia-Manero G, et al. Rapid Donor Identification Improves Survival in High-Risk First-Remission Patients With Acute Myeloid Leukemia. JCO Oncology Practice 2020; 16:6, e464-e475. Access.
- Pidala J, Lee SJ, Ahn KW, et al. Nonpermissive HLA-DPB1 mismatch increases mortality after myeloablative unrelated allogeneic hematopoietic cell transplantation. Blood. 2014; 124(16): 2596-2606. Access.