Researchers at the Fred Hutchinson Cancer Center conducted this retrospective, observational study to compare cytomegalovirus (CMV) reactivation and disease among seropositive recipients of hematopoietic cell transplantation (HCT) using various methods of graft-versus-host disease (GVHD) prophylaxis. Results showed that post-transplant cyclophosphamide (PTCy) in combination with a calcineurin inhibitor (CNI) and mycophenolate mofetil (MMF) was associated with a higher risk of early CMV reactivation when compared to the combination of a CNI with methotrexate (MTX). However, the risk of developing CMV disease was similar across the different study groups, though GVHD ultimately makes patients more susceptible to CMV. This highlights that GVHD prevention remains a priority.
Background
CMV is a common, prevalent virus carried by a significant portion of the population. In individuals with compromised immune systems, such as transplant recipients, CMV can reactivate and lead to serious complications, including an increased mortality risk. The choice of GVHD prevention strategy during transplantation may play a role in the likelihood of CMV reactivation. Understanding the risk of CMV reactivation associated with different GVHD prevention strategies is critical for improving post-transplant outcomes, including survival and the occurrence of GVHD. The objective of this study was to investigate the risk of CMV reactivation using various GVHD prevention strategies to evaluate their impact on important post-transplant outcomes.
Study Details
This study included CMV-positive patients (n=780) who underwent their first allogeneic matched sibling or matched unrelated donor peripheral blood stem cell (PBSC) HCT for hematologic malignancies. The patients were divided into different groups based on GVHD prophylaxis regimen: CNI and MTX (N=322), CNI and MMF (N=414), and CNI and PTCy (N=44). The study occurred from July 2007 to September 2018. It’s important to note that the study was conducted before the standard use of the CMV prophylaxis treatment letermovir.
Results
The study showed that PTCy and the combination of CNI and MMF were associated with a higher risk of early CMV reactivation, within 100 days post-transplant, compared to CNI and MTX. However, the risk of developing CMV disease was similar across the different prophylaxis groups, as shown in the figure below. No significant association was observed between the GVHD prophylaxis approach and the risk of chronic GVHD related to CMV reactivation. Despite the increased risk of early CMV reactivation with PTCy, the risk of CMV disease within 1 year was low in HLA-matched PBSC HCT recipients. The CNI and MMF combination was associated with a higher overall CMV viral burden in the first year post-HCT. While different GVHD prevention strategies may impact CMV reactivation after HCT, effective prevention of GVHD can help reduce the need for corticosteroid treatment and decrease the risk of infections over time.
Key Takeaways
The choice of GVHD prophylaxis regimen can influence the risk of early CMV reactivation in CMV seropositive transplant recipients at greater risk of mortality. While MMF was associated with a higher overall CMV viral burden in the first year post-transplant, the risk of developing CMV disease did not significantly differ across the prophylaxis groups. In addition, no association was found between the GVHD prophylaxis approach and CMV reactivation for chronic GVHD risk. Effective prevention of GVHD remains essential as it can help reduce corticosteroid exposure and mitigate infection risks over time, including CMV reactivation and disease.
Figure
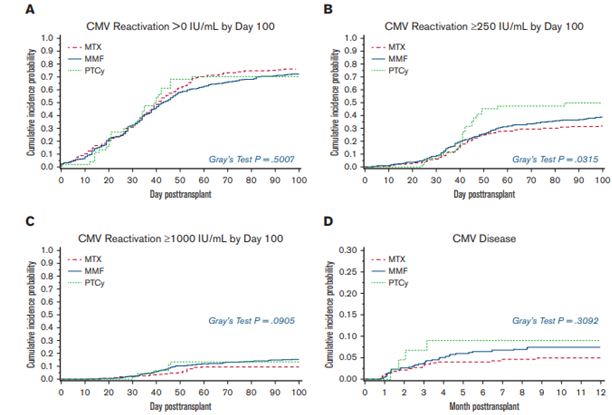
Oshima MU, et al. – Published in Blood Advances
