A study conducted by the CIBMTR® (Center for International Blood and Marrow Transplant Research®) aimed to address the lack of data guiding the selection of haploidentical (haplo) donors for pediatric hematopoietic cell transplantation (HCT). The findings of the study presented at the European Society for Blood and Marrow Transplantation (EBMT) 49th Annual Meeting revealed that sibling donors exhibited a reduced risk of acute and chronic graft-versus-host disease (aGVHD and cGVHD) and graft failure compared to parental donors. Based on these results, the study suggests that choosing sibling haplo donors could be the optimal choice for pediatric HCT over parental donors, leading to minimized risks and improved outcomes.
Background
The selection of donors for HCT correlates with the complications and outcomes of the procedure. Haplo donors, including immediate family members, have become more common in pediatric HCT. However, there is a lack of data to assist pediatric physicians in determining the best haplo donor when multiple options are available. In this study, the researchers hypothesized that using younger age or sibling donors would result in lower incidences of aGVHD and cGVHD.
Study Details
The study analyzed data from 1,069 pediatric patients (≤18 years old) who underwent haplo-related donor transplants for various diseases between 2013 and 2019, as reported to the CIBMTR. The majority of these patients (58%) received transplants to treat malignant diseases, and most (67%) underwent myeloablative conditioning. Post-transplant GVHD prophylaxis strategies included post-transplant cyclophosphamide (PTCy) in 36% of cases, ex-vivo T-cell depletion in 57% of cases, and other approaches in 7% of cases. The analysis divided the donors into 3 age groups: less than 18 years (n=138), 18-35 years (n=440), and greater than 35 years (n=491). Donors included mothers (n=382, median age 36), fathers (n=386, median age 39), and siblings (n=244, median age 16).
Results
The frequency of grade II-IV aGVHD in recipients at 100-days post-HCT was highest with donors greater than 35 years (31%) compared to donors 18-35 (23%) and less than 18 years (15%). Similarly, the incidence of cGVHD at 2 years was also higher with donors greater than 35 years (30%) compared to those 18-35 years (26%) or less than 18 years (16%). Multivariable analysis confirmed that donors older than 35 years had an increased risk of aGVHD compared to donors aged 18-35. There was an increased risk of cGVHD with donors greater than 18 years old, and graft failure was highest with donors 18 years or older (22% for 18-35, 13% for older than 35) compared to donors less than 18 years old (10%). However, there were no differences in severe aGVHD, relapse, or survival based on donor age.
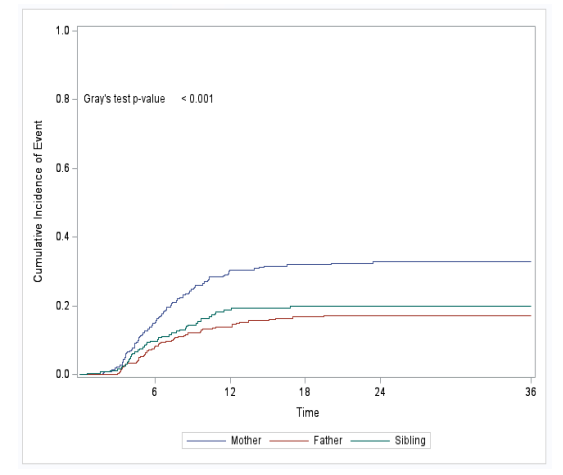
Using parental donors increased the risk of grade II-IV aGVHD compared to sibling donors. Specifically, using mothers as donors significantly increased the risk of cGVHD compared to fathers and siblings, as shown in the figure below. However, no differences in severe aGVHD, relapse, or survival were found based on donor relationship.
Key Takeaways
When selecting haplo donors for pediatric HCT, prioritizing sibling donors can offer significant advantages for patients. This approach helps minimize the risks of aGVHD, cGVHD, and graft failure. This study provides valuable insights, highlighting that sibling donors exhibit lower GVHD risks than older parental donors. These findings fill data gaps to guide the selection and prioritization of haplo donors for pediatric HCT patients. The study suggests the preference for younger donors, such as siblings, may achieve better transplantation outcomes. Findings align with previous studies noting that younger donors are preferable for adult HCT patients. Therefore, considering sibling donors as optimal for pediatric haplo HCT could minimize GVHD and graft failure risks.
Figure: Incidence of cGVHD by Donor Relationship
Liberio N, et al, EBMT abstract pending publication in Bone Marrow Transplantation
